 Full list of publications related to the PhotoCathode ERC CoG projectFuture energy supply depends on innovative breakthroughs to design efficient systems to convert and store the most available source of renewable energy, solar energy. Hydrogen production, based on direct sunlight-driven water splitting in a Photo-Electro-Chemical (PEC) cell, has emerged as a promising and appealing solution. However, such cells need to combine three main characteristics: sustainability, cost-effectiveness and stability. Fulfilling these requirements raises important scientific questions, mainly regarding the development and combination of the best possible materials to harvest light and catalyzee H2 and O2 evolution. Full list of publications related to the PhotoCathode ERC CoG projectFuture energy supply depends on innovative breakthroughs to design efficient systems to convert and store the most available source of renewable energy, solar energy. Hydrogen production, based on direct sunlight-driven water splitting in a Photo-Electro-Chemical (PEC) cell, has emerged as a promising and appealing solution. However, such cells need to combine three main characteristics: sustainability, cost-effectiveness and stability. Fulfilling these requirements raises important scientific questions, mainly regarding the development and combination of the best possible materials to harvest light and catalyzee H2 and O2 evolution.
The objective of PhotoCatH2ode was to design operating photocathodes based on Earth-abundant elements to allow PEC-mediated hydrogen production which would respond to the sustainability and cost-effectiveness issues. The novelty of the project stemmed from the approach, combining organic and hybrid photovoltaics with artificial photosynthesis to design novel materials and architectures. More precisely, I combined and immobilized molecular photosensitizers with bioinspired catalysts on electrode surfaces thanks to electronic junctions. This allowed me to (i) optimize the light-driven charge separation, (ii) control the successive electron transfer steps from the electrode to the catalyst, and (iii) limit charge recombination processes.
|
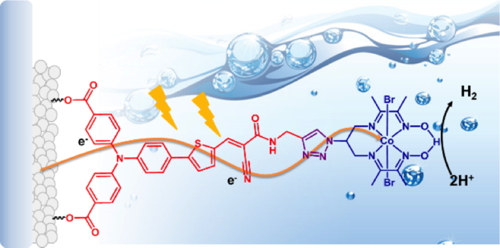
 Molecular photosensitizers were shown to be active in two types of photovoltaic devices: dye-sensitized solar cells (DSSCs, or Grätzel cells) and organic photovoltaic cells. Extending from DSSC technology, I developed a series of molecular photocathodes allowing H2 evolution in aqueous electrolytes. Their design involved the combination of H2-evolving catalysts with donor-acceptor dyes immobilized on p-type semi-conductors. During PhotoCatH2ode, we selected optimal NiO photoelectrode supports,1 designed novel dyes,1a,
2 produced the first all-molecular active photocathode not relying on noble metals (J. Am. Chem. Soc., 2016, Figure 1)3 and optimized its property through molecular design.4 Molecular photosensitizers were shown to be active in two types of photovoltaic devices: dye-sensitized solar cells (DSSCs, or Grätzel cells) and organic photovoltaic cells. Extending from DSSC technology, I developed a series of molecular photocathodes allowing H2 evolution in aqueous electrolytes. Their design involved the combination of H2-evolving catalysts with donor-acceptor dyes immobilized on p-type semi-conductors. During PhotoCatH2ode, we selected optimal NiO photoelectrode supports,1 designed novel dyes,1a,
2 produced the first all-molecular active photocathode not relying on noble metals (J. Am. Chem. Soc., 2016, Figure 1)3 and optimized its property through molecular design.4
|

 In collaboration with Bruno Jousselme (CEA-Saclay), I engineered H2-evolving electrode s by combining organic photovoltaic materials with amorphous molybdenum sulfide.5 This yielded a series of highly active photocathode architectures6 with current densities of several mA.cm–2 and photocurrent onset potential of up to 0.7 V
vs. RHE, which corresponds to more than 50% of the photovoltage required to split water (1.23 V) (Energy Environ. Sci., 2013,
Nanotechnology, 2016). In collaboration with Bruno Jousselme (CEA-Saclay), I engineered H2-evolving electrode s by combining organic photovoltaic materials with amorphous molybdenum sulfide.5 This yielded a series of highly active photocathode architectures6 with current densities of several mA.cm–2 and photocurrent onset potential of up to 0.7 V
vs. RHE, which corresponds to more than 50% of the photovoltage required to split water (1.23 V) (Energy Environ. Sci., 2013,
Nanotechnology, 2016).
|
These novel photo-electrocatalytic systems have now entered a phase where time-resolved photophysical investigations will provide more insights into the sequence of light-driven electron transfer steps that support catalysis.2bIn operando X-ray absorption spectroscopic techniques were used to study the light-driven catalytic mechanism.7 These studies allowed us to demonstrate that polyoxometallates have a significant effect in promoting catalysis through charge photoaccumulation (Energy Environ. Sci.,2013).8
To further enhance catalysis, we designed new catalyst structures, incorporating redox-active moieties or non-innocent redox ligands, and developed novel methods to benchmark the performances of H2-evolving molecular catalysts (Energy Environ. Sci., 2014).9 As highlighted below, this research direction produced unexpected findings, including a novel method to mature hydrogenase enzymes,10 the discovery of an unforeseen polymer coordination structure for amorphous MoSx5, and the design of the most active NiFe mimic of the active site of [NiFe]-hydrogenase known so far.11
|
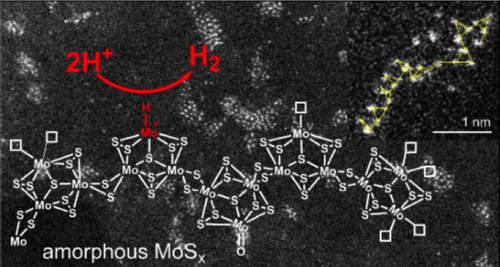
 In collaboration with Dr. Phong D. Tran (Nanyang Technological University, now in the French-Vietnamese University of Hanoi, USTH), we recently used a series of electrochemical, spectroscopic and microscopic techniques to demonstrate that amorphous MoSx is in fact a coordination polymer based on {Mo3S11} clusters and that the catalytic mechanism for H2 evolution involves a MoV-H intermediate (Nature Materials, 2016). This recent study nicely links molecular and solid-state approaches for catalytic H2 evolution and places coordination chemistry at the forefront of the field of solar fuel research.5 In collaboration with Dr. Phong D. Tran (Nanyang Technological University, now in the French-Vietnamese University of Hanoi, USTH), we recently used a series of electrochemical, spectroscopic and microscopic techniques to demonstrate that amorphous MoSx is in fact a coordination polymer based on {Mo3S11} clusters and that the catalytic mechanism for H2 evolution involves a MoV-H intermediate (Nature Materials, 2016). This recent study nicely links molecular and solid-state approaches for catalytic H2 evolution and places coordination chemistry at the forefront of the field of solar fuel research.5
|
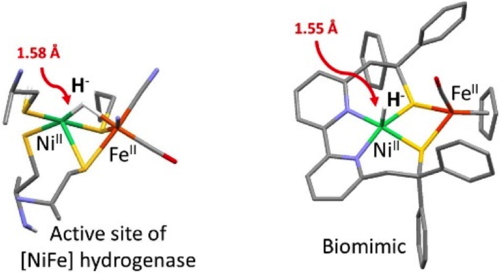
 In collaboration with Carole Duboc (Univ Grenoble Alpes), we were able to develop the first NiFe derivative displaying Ni-centered catalytic reactivity,
i.e. producing H2 thanks to a nickel-hydride intermediate (Nature Chemistry, 2016). Importantly, this catalyst was characterized in four redox states, paralleling those identified with [NiFe]-hydrogenase enzymes.11 In collaboration with Carole Duboc (Univ Grenoble Alpes), we were able to develop the first NiFe derivative displaying Ni-centered catalytic reactivity,
i.e. producing H2 thanks to a nickel-hydride intermediate (Nature Chemistry, 2016). Importantly, this catalyst was characterized in four redox states, paralleling those identified with [NiFe]-hydrogenase enzymes.11
|

 Exploitation of hydrogenases in technological devices requires solutions to be found to limit their O2-sensitivity and to reduce the complexity of their maturation. In 2013, a major breakthrough was achieved by our group thanks to a bioinspired strategy that greatly simplifies the maturation process (Nature, 2013). Apo-hydrogenases can be activated by synthetic di-iron biomimics, with or without the assistance of the protein HydF. This opens fascinating possibilities to study the potential of hydrogenases in biotechnological applications.10 This novel process is also compatible with preparing hydrogenases with labeled active sites, which is helpful when seeking to elucidate the processes involved during maturation of these sites.
Exploitation of hydrogenases in technological devices requires solutions to be found to limit their O2-sensitivity and to reduce the complexity of their maturation. In 2013, a major breakthrough was achieved by our group thanks to a bioinspired strategy that greatly simplifies the maturation process (Nature, 2013). Apo-hydrogenases can be activated by synthetic di-iron biomimics, with or without the assistance of the protein HydF. This opens fascinating possibilities to study the potential of hydrogenases in biotechnological applications.10 This novel process is also compatible with preparing hydrogenases with labeled active sites, which is helpful when seeking to elucidate the processes involved during maturation of these sites.
|