Sandrine Ollagnier & Olivier Hamelin
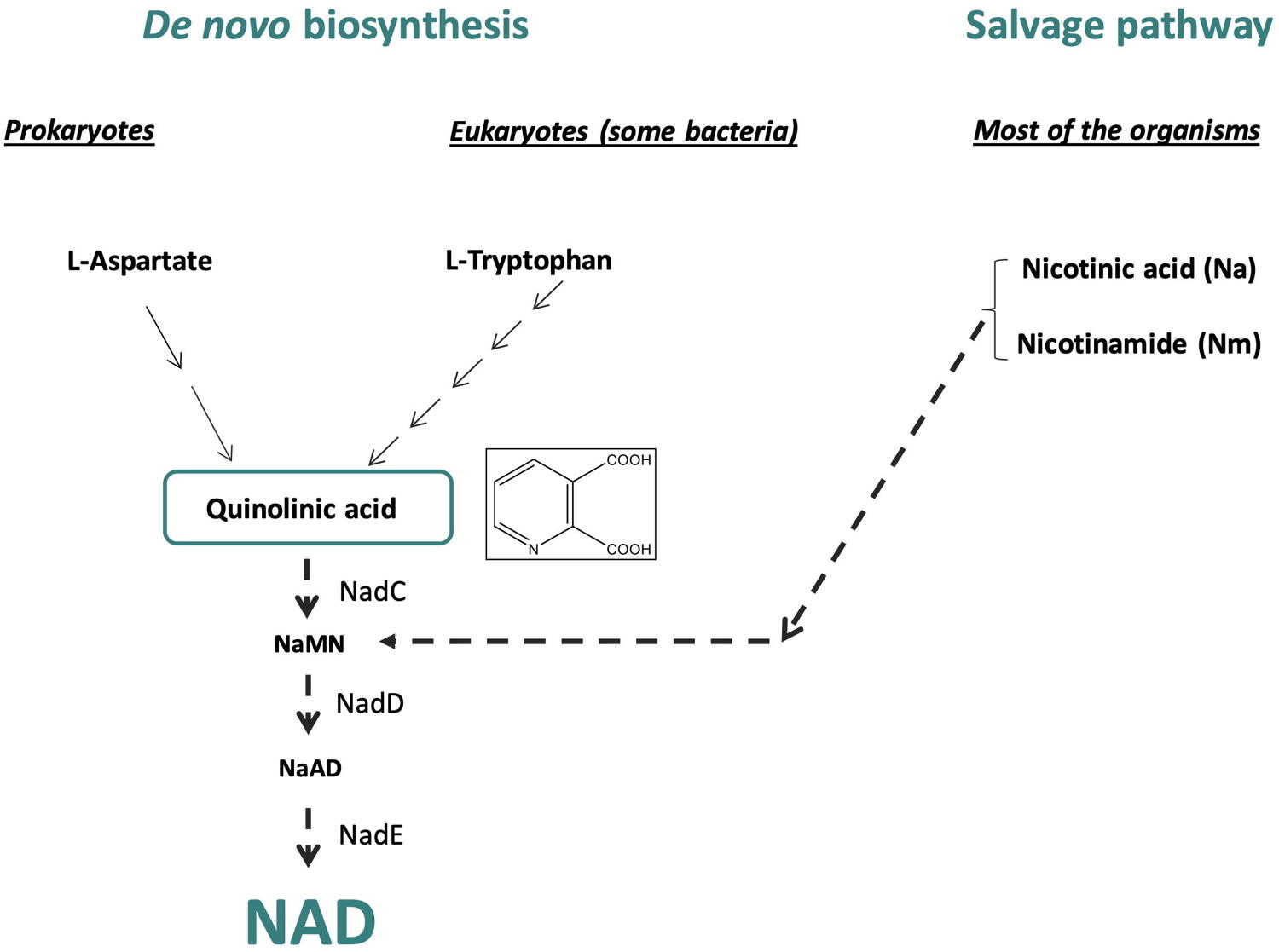
Nicotinamide adenine dinucleotide (NAD) biosynthesis pathway constitutes an interesting metabolic pathway for the discovery of new antibacterial agents. Its biosynthesis involves the formation of quinolinic acid (QA), the biosynthesis of which is different in prokaryotes and eukaryotes. In eukaryotic organisms, QA is produced by the degradation of L-tryptophan, whereas in most prokaryotes, QA is generated from L-aspartate through the concerted action of two enzymes, the L-aspartate oxidase (NadB) and the quinolinate synthase (NadA), a [4Fe-4S] enzyme. Besides these de novo syntheses of NAD, a salvage pathway exists in some organisms that enable NAD to be recycled. However, some pathogens such as
Mycobacterium leprae and
Helicobacter pylori were reported to lack this salvage pathway. This makes NadA a target for the development of antibacterial agents.
We use mainly rational design (substrate and intermediate analogues) to find NadA inhibitors, in collaboration with chemists and virtual screening based on the NadA X-ray structure.