Isabelle Michaud-Soret
Ergothioneine (EGT) is a fascinating small-molecular antioxidant
[1]. Only fungi and bacteria are able to produce it, eukaryotes adsorb it through the diets. There is a growing interest in EGT for its potential health benefits and its role in aging, virulence, dementia and preeclampsia. The literature suggests that the primary role of EGT may be more specific than antioxidant, playing a cofactor role in some enzymes. Indeed the unique chemical functions building EGT: the trimethyl ammonium coupled to a carboxylate and the thione-imidazol group suggest a role for specific interactions. (Figure 1)
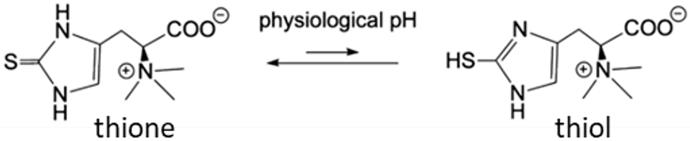 Figure 1
Figure 1: Tautomers of the Ergothioneine structure.
In Preeclampsia, the most threatening pathology of pregnancy, oxidative stress in trophoblast, mitochondrial dysfunction and iron homeostasis disruption were pointed out. EGT may chelate Cu
+/2+/Z
n2+/Co
2+, but, surprisingly, the interaction with Fe
2+/3+ ions, has never been described. It will be crucial to study it in the context of the oxidative stress and air sensitive iron-sulfur cluster. Our project aims to provide new knowledge on the physiological targets of EGT, including iron-sulfur cluster containing proteins or yet unidentified targets and to pave the way for pharmaceutical developments such as antibacterial agents or a biosensor development.
The work is organized in several tasks:
1) the search for specific targets that can bind EGT using specifically designed affinity columns and cell extracts from
Mycobacterium smegmatis, red blood and trophoblast cells;
2) to test the role of EGT on stability of iron/iron-sulfur clusters containing metalloproteins;
3) to produce EGT biosynthesis enzymes from
Mycobacterium smegmatis to test EGT derivatives on their activity in a prospective antibacterial strategy and to
Anabaena cyanobacteria;
4) to demonstrate the proof of concept of a biosensor of EGT in collaboration with a start-up
[1] Cheah IK, Halliwell B (2021) Ergothioneine, recent developments,
Redox biol.
42, 101868.