Anabaena PCC7120 is a cyanobacteria able to fix nitrogen in differentiated cells called heterocysts. In this way, nitrogenase is protected from photosynthetic oxygen evolution performed in vegetative cells. Both types of cells require specific metal ions localization and concentration. To gain insight into the processes of metal biouptake, we propose to perform nano X-ray fluorescence (nanoXRF) imaging on several
Anabaena PCC7120 variants exposed to two different nitrogen conditions. The first aim is to reveal the distribution and concentration of trace metals (Fe, Ni, Mn, Co, Zn) in vegetative and heterocyst cells in environmentally relevant conditions.
Anabaena PCC7120 is a biotechnologically interesting filamentous cyanobacterium able to perform photosynthesis, fix N
2 and form biofilms. The metal and redox homeostasis are tightly controlled to preserve the photosynthetic machinery of vegetative cells and the nitrogen fixation of heterocyst from mismetallation and oxidative cell damage. Vegetative cell differentiation into heterocysts (see figure where vegetative cells
vs heterocysts are shown in green and blue, respectively) only occurs in low nitrate conditions, when N
2 fixation is needed. N
2 fixation is a complicate process relying on the O
2 sensitive nitrogenase requiring compartmentalisation in two types of cells, one producing O
2, the other sensitive to it. To protect the nitrogenase against O
2, heterocysts display no photosystem II activity, enhanced respiration rates and are surrounded by two extra layers of exopolysaccharides and glycolipids
[1]. Because their different electron-transport pathways and the high metal requirements of nitrogenase, both types of cells require specific metal uptake and homeostasis.
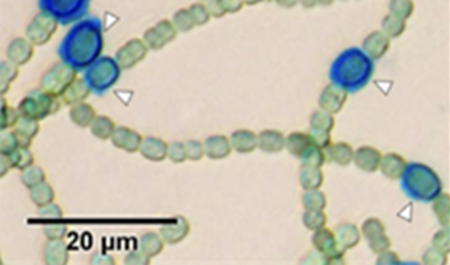 Figure 1
Figure 1: Optical microscopy image of
Anabaena PCC 7120 filaments after 72 hours of nitrate step-down.
Heterocyst are visualized by Alcian blue staining
[2]. Cells in green are vegetative cells.
FUR family members such as FurA, FurB and FurC mainly do the control of this homeostasis.
Ferric uptake regulator (FUR) proteins are prokaryotic transcriptional regulators, which control genes involved in the response to different stresses, including iron, zinc or manganese deficiencies. FurB from
Anabaena sp. PCC7120 works as a zinc uptake regulator (Zur) , while FurC is a peroxide-response regulator (PerR), which also controls the assembly of the photosynthetic machinery and the development and maturation of heterocysts.

Our main goals:

a -
Study the functional and structural properties of these metalloregulators, especially FurA and FurC that control iron homeostasis and stress oxidative response. The role of a newly characterize FurC oligomerization in its transcriptional regulatory activity has been studied by size exclusion chromatography coupled to light multi-angle laser light scaterring and refractometry (SEC-MALLS-RI) experiments (Figure 2. We decipher an additional layer of regulation by reversible oligomerization of FurC (PerR) oxidation dependent, involving the formation of an inter subunit disulfide bridge) Figure 3
[3].
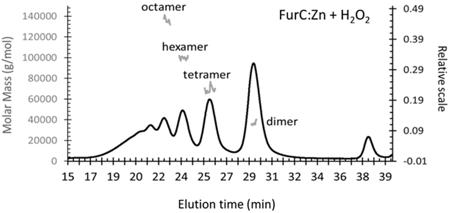 Figure 2:
Figure 2: Molar mass determination by SEC-MALLS-RI of the eluted fraction of the H
20
2 treated FurC: Zn sample showing the presence of well-defined oligomers. The UV at 280 nm profile was shown together with the gray traces corresponding to the calculated Log Molar mass values.
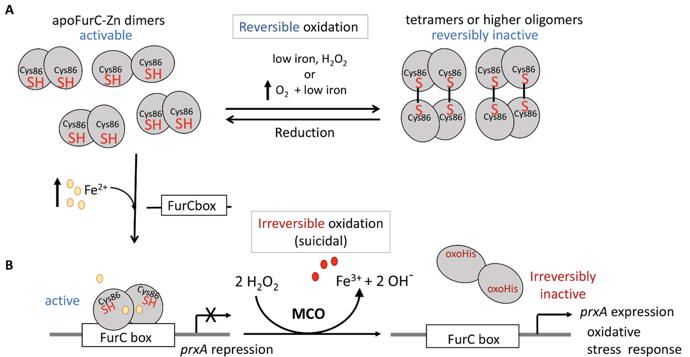 Figure 3
Figure 3: Integrative model of the two levels of FurC oxidation.
A - Reversible intermolecular disulfide bridge formation between FurC dimers under mild oxidant conditions or iron deficiency.
B - Under stronger oxidative conditions caused either by iron repletion and O
2 or Fe + 2 and H
2O
2 , irreversible metal catalyzed oxidation of FurC is produced.

b -
Characterization of the FurC-DNA complex and its sensitivity to oxidative stress by H2O2 and O2. Using structural and biochemical characterizations of the complex stability by SEC-MALS-RI and Electromobility Shift Assays (EMSA).

c -
Visualize and quantify the metal ions distribution in intracellular structures and their spatial organization inside vegetative cells and heterocyst. The morphology of
Anabaena has been studied by microscopy, but the metals concentration and localization are still unknown. To better understand the mechanisms underlying the biological functions of cells related to metal homeostasis, it is crucial to visualize and quantify the metal ions distribution in intracellular structures and their spatial organization inside cells without any modification. This information can be provided by synchrotron nano-probe X Ray Fluorescence (XRF) imaging at the ID16 ESRF Synchrotron beamline in collaboration with G. Veronesi (ComX group at LCBM) as previsiously performed
[4]. On the other hand, unlike biofilms of other microorganisms, cyanobacterial biofilms contain negatively charged exopolysaccharides (EPSs) able to adsorb metals. Visualization and metal distribution in the biofilm will give some clues for potential applications of cyanobacteria in heavy metal chelation for detoxification purposes.
[1] Pivotal Role of Iron in the Regulation of Cyanobacterial Electron Transport. A González, E Sevilla, MT Bes, ML Peleato, MF Fillat.
Advances in Microbial Physiology, Chapter 5. Editor(s): Robert K. Poole, Academic Press,
68, 2016, Pages 169-217, ISSN 0065-2911, ISBN 9780128048238. doi: 10.1016/bs.ampbs.2016.02.005
[2] FurC (PerR) from Anabaena sp. PCC7120: a versatile transcriptional regulator engaged in the regulatory network of heterocyst development and nitrogen fixation. C Sarasa-Buisan, J Guio, E Broset, ML Peleato, MF. Fillat and E Sevilla.
Environmental Microbiology (2022)
24(2), 566-582
[3] Metal binding and oligomerization properties of FurC (PerR) from Anabaena sp. PCC7120: an additional layer of regulation? C Sarasa-Buisan, E Emonot, M Martínez-Júlvez. E Sevilla, A Velázquez-Campoy, S Crouzy, MT Bes, I Michaud-Soret, MF Fillat.
Metallomics, 2022, Oct 6. doi:10.1093/mtomcs/mfac077
[4] Visualization, quantification and coordination of Ag(+) ions released from silver nanoparticles in hepatocytes. G Veronesi, A Deniaud, T Gallon, PH Jouneau, J Villanova, P Delangle, M Carriere, I Kieffer, P Charbonnier, E Mintz, I Michaud-Soret.
Nanoscale 2016,
8 (38), 17012-17021. doi: 10.1039/c6nr04381j