Research directed at developing hydrogen production techniques is largely based on observations of the chemical reactions that take place during natural photosynthesis. Under specific conditions, micro-organisms such as algae and bacteria use light energy to produce hydrogen from water. In order to reproduce and adapt these processes, researchers have developed molecular systems capable of both capturing light energy (photosensitization) and using the energy collected to liberate hydrogen from water (catalysis). All technological systems developed to produce or use hydrogen have so far relied on the use of noble metals such as platinum [1], but this has one significant drawback - the metal's limited reserves. Despite all the efforts to reduce the quantities of platinum used in electrolysers and batteries, the metal's scarcity and cost hinder the hydrogen energy sector's long-term economic development. Current research is focused on finding ways to bypass the use of platinum, in particular by developing catalysts based on metals that are naturally more abundant, and therefore more economical, such as those used by natural organisms i.e. iron, nickel, cobalt and manganese.
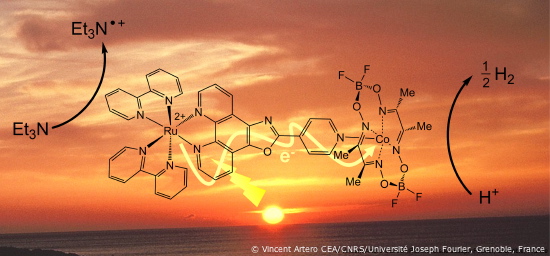
The recently developed system is a supramolecular device where the photosensitization and catalysis functions are performed by one and the same molecule. Under light irradiation, a ruthenium center accepts electrons from an organic molecule and drives them to the cobalt catalyst which evolves hydrogen. This system demonstrates greater efficiency than comparable systems using noble metal catalysts (Pd, Rh and Pt) described above. A next step will consist of substituting a cheaper photosentitizer for the ruthenium center.
While the long-term objective is to use water as a source of both protons and electrons (thus avoiding the use of a sacrificial organic molecule), the published findings represent a major step forward in the field of hydrogen photoproduction.
[1] Historically, noble metals meant precious metals such as gold, silver, platinum, etc. traditionally used in jewellery design. Chemists, however, use the term to describe metals that are resistant to oxidation. Nowadays, this term also applies to metals such as palladium, rhodium, ruthenium or iridium whose particularly low terrestrial abundance explains both their scarcity and high cost.