Our findings
Published on 25 January 2019
Body text 1
 Metal transport site of P-type ATPases: the primary determinant of ion selectivity of the transporter
Drawing from the work done on SERCA1a, the Ca(II)-ATPase of the sarcoplasmic reticulum (Figure 3) and among the best known P-type ATPase, we seek to understand how a transition metal crosses the membrane through the transporter.
Metal transport site of P-type ATPases: the primary determinant of ion selectivity of the transporter
Drawing from the work done on SERCA1a, the Ca(II)-ATPase of the sarcoplasmic reticulum (Figure 3) and among the best known P-type ATPase, we seek to understand how a transition metal crosses the membrane through the transporter.
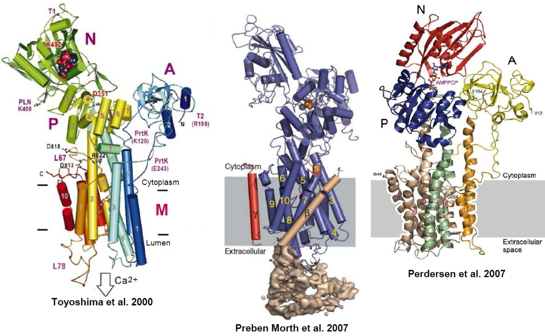
Figure 3: Images of the three dimensional structures of P-type ATPases resolved to date. From left to right: Ca2+-ATPase SERCA1a; Na+/K+-ATPase; H+-ATPase.
Combining, molecular biology, heterologous expression and functional analysis by biochemical methods [Bal et al., 2001; Bal et al., 2003), our work has highlighted the critical and distinct roles of the two cysteine of the sixth membrane helix of Ccc2 and CadA, and specified the amino acid involved in Cd(II) transport sites [Lowe et al., 2004; Wu et al., 2004; Wu et al., 2006a]. This work is continuing with CadA including the development of a structural component in collaboration with the laboratory of Thomas Sorensen in Oxford (UK).
 Regulating the activity of ATPases-Cu (I) by phosphorylation
One feature of Cu(I)-ATPases of eukaryotes is their regulation by kinases and phosphatases, proteins that alternating phosphorylation and dephosphorylation of certain amino acids of the Cu(I)-ATPases modulate the speed of the transport cycle. We study the role of serine may be sites of action of kinases and phosphatases in the functioning of Ccc2 to clarify regulations that may occur in the cell. This work is made in collaboration with the laboratory of Professor A. Vieyra in Rio de Janeiro [Valverde et al., 2008; Valverde et al., 2010].
Regulating the activity of ATPases-Cu (I) by phosphorylation
One feature of Cu(I)-ATPases of eukaryotes is their regulation by kinases and phosphatases, proteins that alternating phosphorylation and dephosphorylation of certain amino acids of the Cu(I)-ATPases modulate the speed of the transport cycle. We study the role of serine may be sites of action of kinases and phosphatases in the functioning of Ccc2 to clarify regulations that may occur in the cell. This work is made in collaboration with the laboratory of Professor A. Vieyra in Rio de Janeiro [Valverde et al., 2008; Valverde et al., 2010].
 Selectivity and detoxification
Each membrane protein that we study is the specific to the ion it transports; this specificity is more or less strict. For example, CadA can carry Cd(II) but also Zn(II) or Pb(II); Ccc2 can carry Cu(I) but also Ag(I). However, CadA cannot transport Co(II) or Cu(I). As described above, the main determinant of ion selectivity of P-type ATPases is the membrane transport site.
Selectivity and detoxification
Each membrane protein that we study is the specific to the ion it transports; this specificity is more or less strict. For example, CadA can carry Cd(II) but also Zn(II) or Pb(II); Ccc2 can carry Cu(I) but also Ag(I). However, CadA cannot transport Co(II) or Cu(I). As described above, the main determinant of ion selectivity of P-type ATPases is the membrane transport site.
However, a second domain of interaction with the metal, located at the N-terminus of the protein is found on most of the PIB-type ATPases. This domain has a ferredoxin-like structure and a Cys-X-X-Cys consensus sequence (Figure 4). In the cytosol, small proteins called metallochaperones, such Atx1 in yeast, bind Cu(I) by the same consensus sequence. The chaperones and N-terminal domains of PIB-type ATPases constitute a family of proteins having the same fold and the same binding motif of heavy metals, but they set, as appropriate, Cu(I), Cd(II) , Zn(II) or Hg(II) (Figure 4). We focus on these metal binding domains both at a structural and a functional point of view and in particular on their role in the selectivity of the carrier for metals [Morin et al., 2005; Banci et al., 2006; Miras et al., 2008; Poger et al., 2008; Morin et al., 2009].
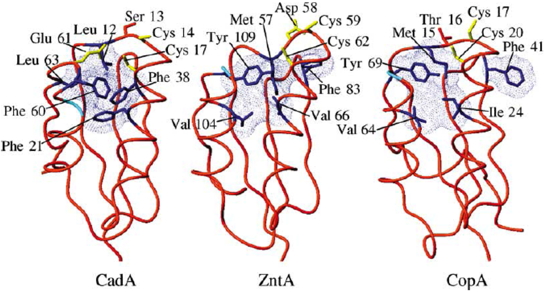
Figure 4: Structure of N-terminal domains of metal binding (MBD) of three P-type ATPases. CadA, Cd(II)-ATPase of L. monocytogenes, ZntA, Zn(II)-ATPase of E. coli, CopA, Cu(I)-ATPase of B. subtilis. Yellow, amino acids involved in metal coordination. Blue, conserved hydrophobic region in the three areas. (Banci et al. 2006)
 The Unfolded Protein Response in yeast is triggered by an exposure to cadmium
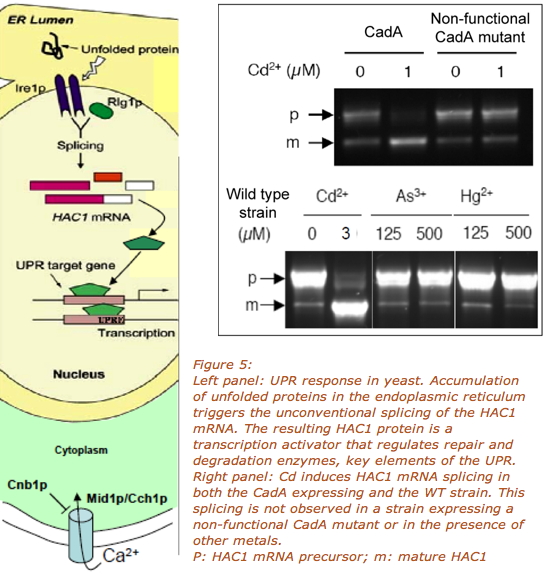
The expression of CadA in S. cerevisiae increases yeast sensitivity to Cd (Wu et al., 2004). To understand the molecular mechanisms sustaining this phenotype, we performed a transcriptomic analysis which revealed that in the presence of the bacterial transporter, a short exposure (1h) to low Cd concentrations (in the micromolar range) triggered the Unfolded Protein Response (UPR). We found that on a WT yeast strain, Cd also induced UPR (Figure 5). This intracellular response to Cd is coupled to an increased calcium entry through the CCH1/MID1 channel located at the plasma membrane. We also evidenced that this channel could behave as a cadmium entry pathway. (Gardarin et al., 2010)
The Unfolded Protein Response in yeast is triggered by an exposure to cadmium
The expression of CadA in S. cerevisiae increases yeast sensitivity to Cd (Wu et al., 2004). To understand the molecular mechanisms sustaining this phenotype, we performed a transcriptomic analysis which revealed that in the presence of the bacterial transporter, a short exposure (1h) to low Cd concentrations (in the micromolar range) triggered the Unfolded Protein Response (UPR). We found that on a WT yeast strain, Cd also induced UPR (Figure 5). This intracellular response to Cd is coupled to an increased calcium entry through the CCH1/MID1 channel located at the plasma membrane. We also evidenced that this channel could behave as a cadmium entry pathway. (Gardarin et al., 2010)
 The PIB-type ATPases of the chloroplast
The chloroplast is a specialized organelle hosting photosynthesis activity and possessing 3 PIB-type ATPases. In the plant Arabidopsis thaliana, two of them PAA1 (or HMA6) and HMA1 are located in the envelope, the third one PAA2 (or HMA8) in the thylakoid membrane (Figure 6). If it exists diverse hypotheses concerning HMA1 function in plant, neither PAA1 nor PAA2 had been biochemically studied so far.
The PIB-type ATPases of the chloroplast
The chloroplast is a specialized organelle hosting photosynthesis activity and possessing 3 PIB-type ATPases. In the plant Arabidopsis thaliana, two of them PAA1 (or HMA6) and HMA1 are located in the envelope, the third one PAA2 (or HMA8) in the thylakoid membrane (Figure 6). If it exists diverse hypotheses concerning HMA1 function in plant, neither PAA1 nor PAA2 had been biochemically studied so far.
On PAA1 produced in the bacterium Lactococcus lactis, we performed phosphorylation assays from ATP or inorganic phosphate. We thereby demonstrated that PAA1 is a Cu+-ATPase, displaying the main enzymatic characteristics of P-type ATPases. We confirmed the ionic selectivity of PAA1 by an original phenotypic test in the yeast S. cerevisiae [Catty et al., 2011].
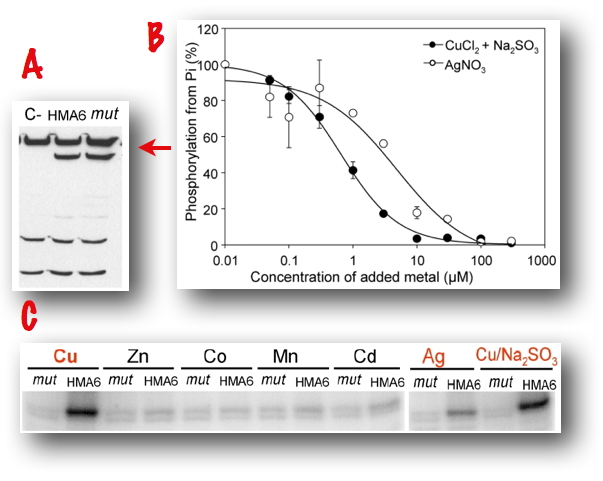
Figure 6: HMA6 is correctly produced in Lactococcus lactis as shown by Western blot analysis (A). In the presence of ATP, HMA6 is transiently phosphorylated in the presence of Cu(I) or Ag(I) at micromolar concentrations. Conversely, Cu(I) and Ag(I) inhibit phosphorylation from inorganic phosphate. mut = phosphorylation site mutant of HMA6.
 New project: Metal homeostasis in chloroplasts: comparative study of three transporters belonging to the PIB-type ATPases family
The aim of this project is to characterize in details the chloroplastic ATPases HMA1, 6 and 8 by both biochemical and structural approaches. To reach this goal, native and mutated forms of the transporters will be first produced in L. lactis. Their enzymatic properties will be further determined by phosphorylation assays previously setup for HMA6 study. The collected informations will allow a comparison of the functional characteristics of the three HMAs in terms of ionic specificity, affinity for metals and ATP and will also reveal aminoacids or domains essential for their activity. In parallel, HMA6 will be purified in an active form and in large amount, prerequisite for the structural study of this transporter.
New project: Metal homeostasis in chloroplasts: comparative study of three transporters belonging to the PIB-type ATPases family
The aim of this project is to characterize in details the chloroplastic ATPases HMA1, 6 and 8 by both biochemical and structural approaches. To reach this goal, native and mutated forms of the transporters will be first produced in L. lactis. Their enzymatic properties will be further determined by phosphorylation assays previously setup for HMA6 study. The collected informations will allow a comparison of the functional characteristics of the three HMAs in terms of ionic specificity, affinity for metals and ATP and will also reveal aminoacids or domains essential for their activity. In parallel, HMA6 will be purified in an active form and in large amount, prerequisite for the structural study of this transporter.
Our results should improve our knowledge of plant PIB-type ATPases and provide new informations on the molecular mechanisms that occur during metal transport by these proteins. Together with the data coming from in planta studies, they should also allow to specify the relative role of HMA1, 6 and 8 in the metal homeostasis of chloroplast.
Top page