Degradation of per- and polyfluoroalkyl substances (PFAS)
Published on 9 February 2026
Body text 1
N. Duraffourg
Scientific project : The degradation of per- and polyfluoroalkyl substances (PFAS)
- Context : Sustainable pollution at the heart of health and environmental concerns
The specific characteristic of PFAS, which comprise several thousand synthetic chemical compounds used in many industrial sectors and everyday products since the mid-20th century, is their extreme chemical stability (dissociation energy = 490 kJ.mol-1). They are not very biodegradable and persist in the environment for a long time, which has earned them the label of ‘forever chemicals’. Their presence is now widely documented in surface and groundwater, soil, ecosystems and the food chain. This widespread distribution leads to almost permanent exposure of populations (1,2,3).
- Project 1 : Sustainable degradation of PFAS by electro-catalysis
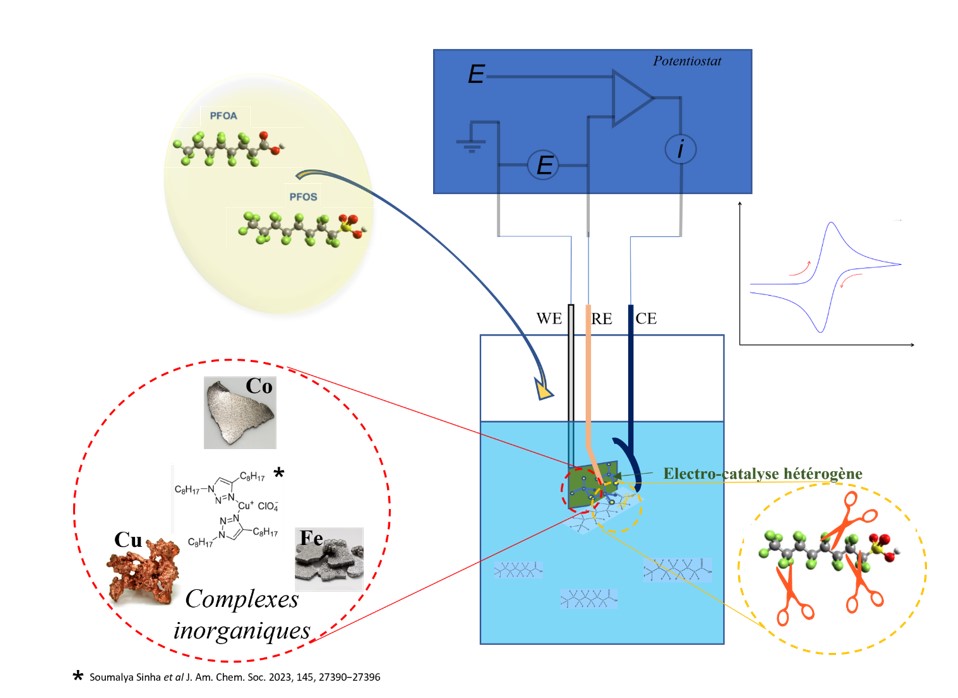
The degradation processes developed to date are sometimes highly effective, but even the most technologically advanced ones require drastic conditions (acidic or basic environments, high temperatures, chemical oxidants, etc.) and consume a lot of energy. We aim to develop the degradation of PFAS by electrocatalysis in an aqueous environment, a gentle process, using inorganic transition metal complexes. A recent study has highlighted a copper-based molecular catalyst that enables rapid degradation of PFOA with high rates of mineralisation and defluorination at room temperature (4). We plan to develop new inorganic complex catalysts based on transition metals (Cu, Co, Fe) for the electrocatalytic degradation of PFAS. We will focus our strategy on electrochemical control of catalyst regeneration and will seek to elucidate the electrocatalytic mechanisms involved.
We base our working hypothesis on the following characteristics:
- Electrocatalysis makes it possible to envisage the degradation of these molecules at the molecular level via controlled electron transfers, often at room temperature.
- Inorganic copper complexes, for example (Cu(I)/Cu(II) stabilised by inorganic ligands), can act as catalytic centres to initiate the breaking of C–C and C–F bonds.
Goal :
-
Complete degradation of PFAS.
An understanding of the electrochemical and chemical mechanisms involved.
-
Anses. (2025). Campagne nationale de mesure de l'occurrence de composés émergents dans les eaux destinées à la consommation humaine : PFAS et US-PFAS - Campagne 2024-2025. Maisons-Alfort : Anses, 60 p.
-
Rapport de l'Académie des sciences – 25 mars 2025 : La pollution aux PFAS : état des lieux des connaissances et enjeux de société.
-
Rapport INERIS Ineris - 229253 - 2808401 - v2.0 - 08/04/2025 : Identification des principales voies d'exposition aux PFAS.
-
Soumalya Sinha et al J. Am. Chem. Soc. 2023, 145, 27390−27396.
- Project 2 : Towards bioremediation of PFAS
The degradation of PFAS within bio-hybrid systems is also being developed in collaboration with
Dr Olivier Hamelin, from the BioCat team at the
CBM laboratory. It has been shown that PFAS intercalate into specific pockets in proteins (5). We wish to exploit this property to develop bio-hybrid catalysts in aqueous phase. Indeed, based on this protein-absorbing property (6), the addition of an exogenous catalyst makes it possible to envisage the development of a hybrid enzyme whose efficiency could be modulated by specific mutation.
5. Y. D. Yang et al Biophysical Chemistry 302 (2023) 107110
6. Environ. Sci.: Water Res. Technol., 2022, 8, 1188–1194
Skills/resources: :
- Inorganic chemistry.
- Electrochemistry.
- Analytical chemistry (NMR, EPR, HPLC-Mass, GC-Mass, UV-Vis and FTIR spectroscopy, ICP-AES).
- Overproduction and purification of proteins of interest.
- Molecular biology.
- Crystallogenesis.
- Structural analysis of biopolymers.
Top page
Top page