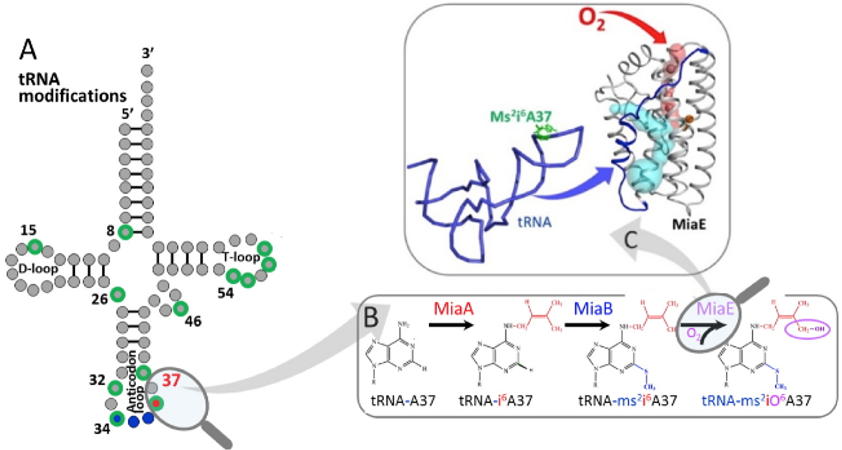
The results demonstrates that the putative
pp-2188 gene encodes a MiaE enzyme. The structure shows that Pp-MiaE consists of a catalytic diiron(III) domain with a four alpha-helix bundle fold. A docking model of Pp-MiaE in complex with tRNA, combined with site directed mutagenesis and
in vivo activity shed light on the importance of an additional linker region for substrate tRNA recognition. Finally, krypton-pressurized Pp-MiaE experiments, revealed the presence of defined O
2 site along a conserved hydrophobic tunnel leading to the diiron active center.