Sandrine Ollagnier de Choudens, Olivier Hamelin, C. Gerez, P. Carpentier, I. Elchennawi
Maturation of metallic sites such as iron-sulfur clusters (Fe-S), canonical or complex ones (containing additional heteroatoms), remains a very active and competitive field in bioinorganic chemistry. Maturation systems are structurally and functionally complex and more and more diseases are revealed due to defects in Fe-S assembly process, pointing to the importance to study at a molecular level such a process.
To date, five Fe-S biogenesis systems that catalyze Fe-S cluster acquisition by apo-proteins have been described, namely NIF, ISC, SUF, MIS and SMS. The BioCatalysis team was pioneer in the study at a molecular level of Fe-S biogenesis by the SUF system in
Escherichia coli. We are working to understand, in collaboration with microbiologists, spectroscopists and structuralists, how the six SUF proteins SufA-SufB-SufC-SufD-SufS-SufE, interact together and control the complex assembly of Fe and S to form Fe-S clusters. Recently, we investigated the SUF-like system from
Mycobacterium tuberculosis, which constitutes an interesting therapeutic target against Tuberculosis. The team is also working on the ISC system, composed by IscS-IscU-IscA-HscA-HscB-Fdx-IscX and CyaY proteins, with a focus on IscS, IscU and CyaY proteins. We have recently initiated also the structural and functional characterization of the ancestral SMS system.
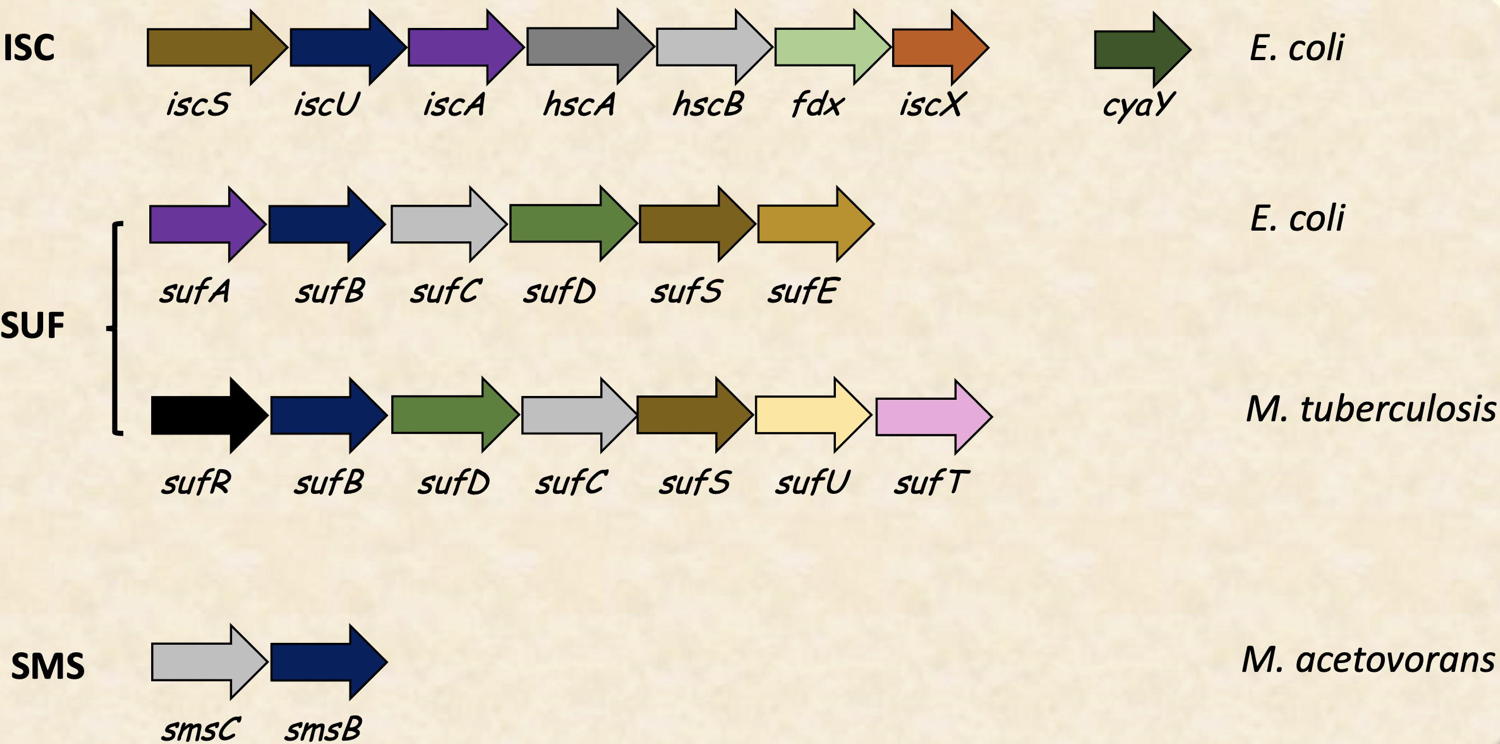
Fe-S biogenesis systems studied in the BioCat team
Grant
ANR BAMBI (ANR-24-CE44-7921) (2024-2027); ANR CovFeS (ANR-24-CE44-1662) (2024-2027); ANRS0522 FeSMtb (2024-2027); ANR-22-CE20-0039 MASTIC (2023-2027); ANR-22-CE44-0043 FirstFeS (2023-2027); GRAL2-CBMTIE (2023-2024).