The chemical industry relies on chemical catalysis to increase the yield and selectivity of the reactions used in its production processes. The industry is also looking into using enzymes, which are natural catalysts. However, these biomolecules are fragile and can only be used for a limited number of reactions. The idea of scientists is to take inspiration from their functional mechanisms, to create artificial customized enzymes following the needs and constraints of industry.
Researchers at our laboratory in collaboration with the IBS (in Grenoble) have mastered, for the first time, a method to construct an artificial metalloenzyme. This enzyme is comprised of a backbone and an active site having an affinity for a reagent,
i.e. the molecule that will be transformed by the chemical reaction. For a backbone, the researchers chose a protein without any activity that is stable in relatively aggressive environments, and contains a pocket in which the active site is located. This site, which is central to the chemical reaction, contains a metallic iron-based molecule. The third and final player is the reaction’s reagent, which must have an affinity with the enzyme. How can the catalytic efficiency of this artificial enzyme be verified? The scientists have sought out eligible reagents for a well-defined chemical reaction (that is also used by the pharmaceutical industry), namely the transfer of an oxygen atom to a sulfur atom. Starting from “molecular docking” calculations, 300 candidate molecules were identified. Tests conducted on some of them show that the reaction does occur at the active site of the artificial enzyme. In addition, the number of catalytic cycles is much greater than that achievable by chemical catalysis.
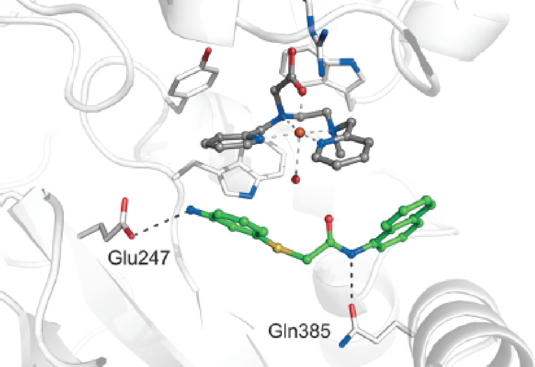
Representation of the position of the substrate chosen for the oxidation of its thioether function by the FeL iron complex within NikA.
The findings represent a new stage in the development of artificial enzymes derived from chemistry and biology, as well as for sustainable chemistry. The first potential application: the development of anti-ulcer compounds, analogs of drugs such as omeprazole.