In the field of synthetic biology, biocatalysis represents the most promising solution for developing sustainable chemistry. However, there is still work to be done, particularly in developing abiotic reactions and improving the stability of biocatalysts. The approach we are developing involves the creation of artificial metalloenzymes (ArMs): bio-inspired inorganic complexes are introduced into the hydrophobic cavities of proteins. The inorganic complex acts as the (artificial) reactive centre, while the protein provides stability and (enantiomeric) selectivity. The aim of our project is to develop hybrid catalysts that are active in oxidation reactions, focusing on asymmetric oxygen transfer reactions on an unlimited quantity of substrate.
 Protein hybrid chiral catalysts: towards heterogeneous and multifunctional artificial metalloenzymes
Protein hybrid chiral catalysts: towards heterogeneous and multifunctional artificial metalloenzymes
In collaboration with BEE et COMMIX teams of the laboratory.
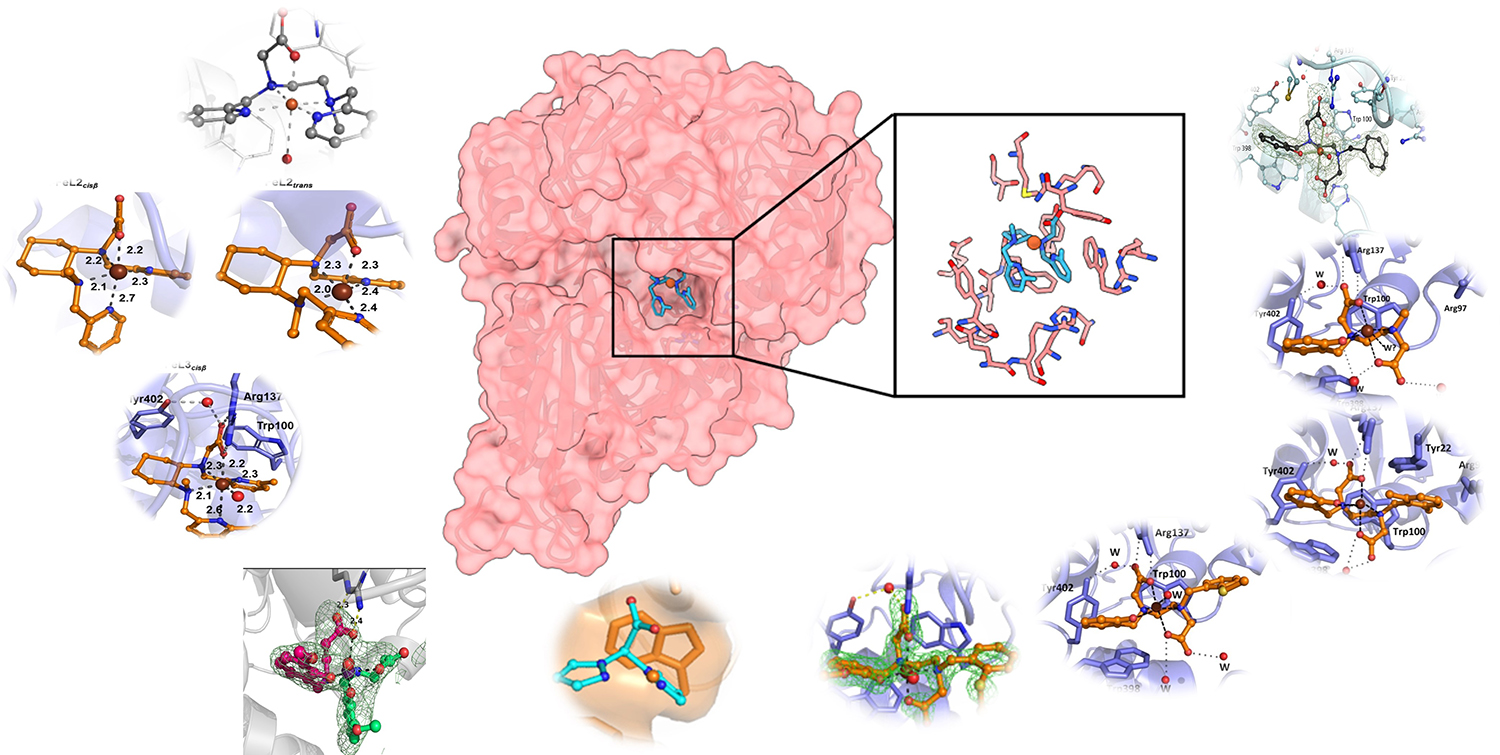
Despite all the progress made in research, it is still difficult today to predict the outcome of enantioselective chemical catalysis. However, these reactions are perfectly controlled by metalloenzymes, due to the nature of the structure of the first and second coordination spheres of the metal within the protein. Our approach aims to draw inspiration from this mode of control to ensure enantioselective oxidation reactions through the development of artificial enzymes. The enzyme chosen is NikA, a Ni transporter in E. coli, and the inorganic complexes inserted into the protein that act as the active site are derived from inorganic catalysis for oxygen or nitrogen transfer and CO2 fixation. Three types of catalysis are then considered and compared, most often for alkene transformations.
In vitro catalysis: Artificial NikA@ML enzymes (with M = Fe, Ru, Mn) enable hydrochlorination and epoxidation reactions in a buffer, whereas the complexes alone are completely inactive. This phenomenon is always observed, demonstrating the importance of protein structure. Given our limited success with enantioselectivity, we decided to use AI to create effective artificial enzymes for sulfoxidation catalysis from scratch, using reduced esomeprazole as a substrate. We have now mastered the methodology and results should follow.
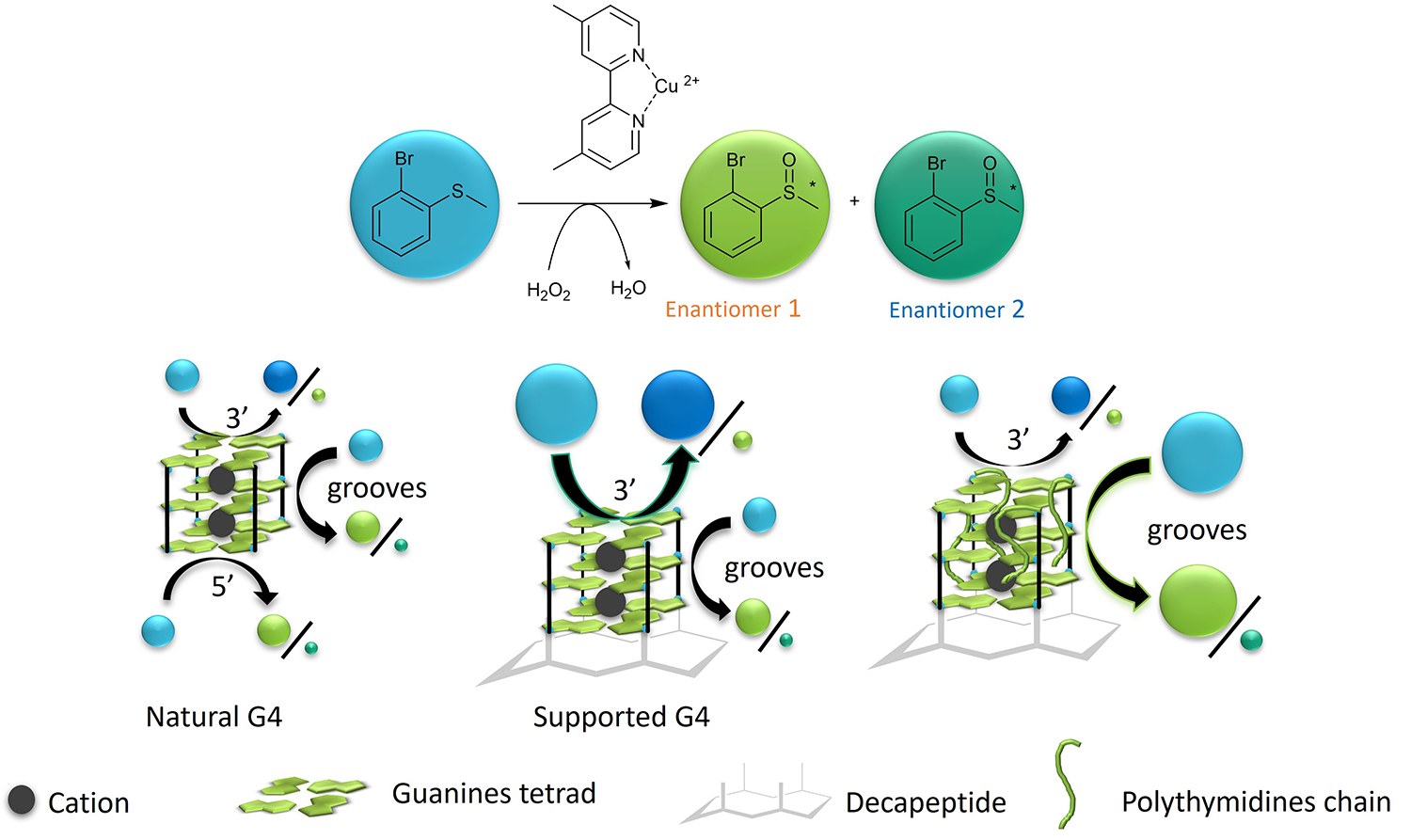
Other biomolecules are currently being tested, such as G-quadruplexes, which have been used for sulfoxidation reactions.
In vivo catalysis
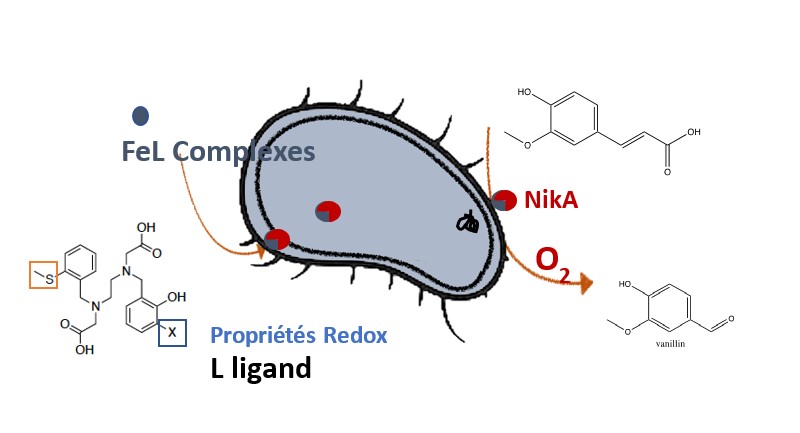
: We are seeking to produce our artificial enzyme directly in
E. coli by adding our complex to bacterial cultures. Here, the added value lies in the continuous production of artificial enzymes, enabling large-scale production of products of interest and therefore greater virtual stability. This work is illustrated by the production of vanillin from lignin residues such as ferulic acid.
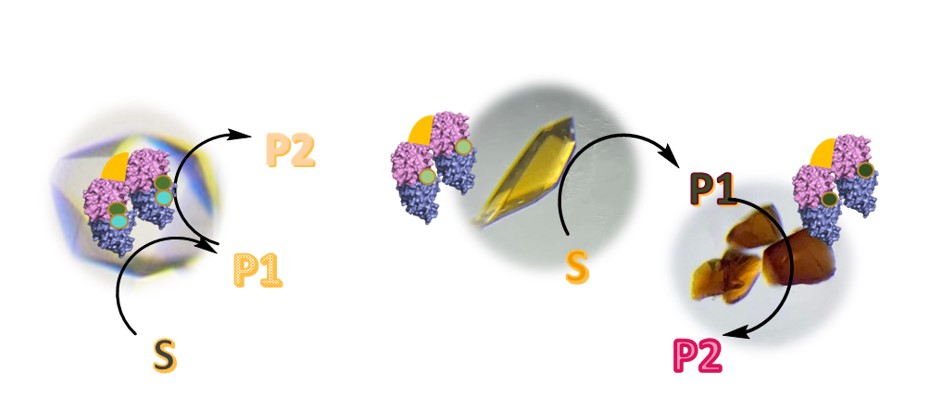
In cristallo catalysis: The aim here is to transpose heterogenisation into the field of biocatalysis through the use of catalytic protein crystals. We have developed heterogeneous hybrid catalysts by stabilising protein crystals using the CLEC (Cross Linked Enzyme Crystals) technique. In these CLECs, inorganic complexes are internalised as required, depending on the target reaction. These heterogeneous catalysts are very stable under oxidative conditions, organic solvents, agitation, temperature and pH. They have enabled us, for example, to carry out an oxidative cleavage reaction of alkenes using oxygen from
the air with a TON greater than 30,000.
Today, we plan to apply these concepts to other reactions such as nitrogen transfer. We are also attempting to develop cascade reactions using CLECs by inserting not one but two artificial active sites, with the aim of avoiding interference between catalysts and increasing selectivity. The formation of inorganic cyclic carbonates from an alkene is achieved by inserting two complexes (Co and Mn) into the same NikA protein crystal using two different methods: stabilisation of the complex by supramolecular bonding as usual, and by covalent bonding through the reaction of a cysteine mutant of the NikA protein with a maleimide function, for example. Another option is to use a mixture of two types of CLEC, each containing a reaction site dedicated to one of the targeted reactions (in this case, Fe and VO complexes).